When Dentsply Sirona’s intraoral scanner Primescan launched in 2019, it could be used for patient consultation, treatment planning in implant dentistry and orthodontics, and for restorative treatments.
Now, dentists can also use the scans in a validated workflow to create custom-made oral appliances, courtesy of Dentsply Sirona’s new partnership with Panthera Dental.
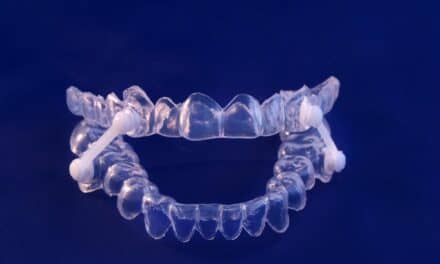
Dentsply Sirona’s Primescan creates a full-arch scan in as little as a minute to give the dentist immediate control of a 3-D model. The Primescan delivers reliable data for the Panthera Dental D-SAD custom-made sleep apnea oral appliance.
For dentists in North America, Germany, France, Belgium, Netherlands, Luxembourg, the United Kingdom, Australia, and New Zealand, the data will be transferred securely via the Dentsply Sirona Connect Case Center. For other Primescan and Panthera Dental countries, the data can be sent via STL to Panthera Dental through their web portal (regulations per country and availability may vary). In a future software version, a full integration in the Connect Case Center for all Primescan and Panthera Dental-validated countries is planned.
[RELATED: Demystifying Intraoral Scanning in Dental Sleep Medicine]
After using Primescan to scan the patient’s complete jaw and the bite registration for the protrusion, the scan data can be sent directly to Panthera Dental, either using the Connect Case Center or via STL-file. Within 15 days, the D-SAD oral appliance will be designed and manufactured by Panthera Dental’s proprietary CAD/CAM process in their 4.0 manufacturing plant using digital, robotic, and automated technologies. It is then shipped back to the dental professional via multinational express delivery service.
Dental sleep medicine educator Shouresh Charkhandeh, PhD, DDS, says in a release, “Over one billion people worldwide suffer from sleep-related breathing disorders such as obstructive sleep apnea. Many of them can benefit from oral appliance therapy (OAT). We need to safely simplify OAT and make it predictable in order for many dentists to get involved in providing this life-saving treatment to patients. The validated digital workflow solution between Primescan and Panthera Dental, which combines Primescan’s impressive accuracy, efficiency and ease of use, with Panthera Dental’s precise and versatile manufacturing, enables dentists around the world to help many suffering patients live a longer, better and healthier life and provide an alternative solution to CPAP therapy.”
Panthera Dental’s D-SAD is made out of biocompatible type-12 polyamide nylon. A wide range of plateaus and bands are available and allow for over 300 standard design combinations as well as maximized tongue space. Additional customization is possible, and the D-SAD can be adapted to every patient morphology, according to Panthera.
The D-SAD’s titration mechanism uses interchangeable rods made of the same biocompatible polymer as the device, designed to not elongate and to resist bruxism. The length of the rods varies from 16 to 34 mm and can be adjusted in 0.5 mm increments for an accurate and customized advancement. A patented connection system ensures the replaceable rods do not disengage during sleep.
“We are fulfilling an explicit request from customers and providing added value for dentists working with Primescan,” says Alexander Völcker, PhD, group vice president CAD/CAM at Dentsply Sirona, in a release. “The combination of Panthera Dental’s Digital Sleep Apnea Devices and Primescan is a great example of what can be achieved when a dental workflow starts with an intraoral scan. The digital technology integrates seamlessly into the daily workflow, enabling clinicians to provide a fast and accurate solution for sleep apnea and snoring.”
Beatrice Robichaud, co-founder and VP marketing & customer experience at Panthera Dental, says in a release, “Our collaboration with Dentsply Sirona is a natural fit that will synergize both our companies’ recent technological advancements in the field of CAD/CAM solutions. Dentists worldwide will benefit from the predictability of the validated Primescan workflow while delivering an accurate, completely customized Digital Sleep Apnea Device to their patients, for greater patient satisfaction and quality of life.”
The Panthera D-SAD received FDA 510(k) clearance for snoring and obstructive sleep apnea has the CE mark and complies with the Canadian Medical Devices Regulations.