The manuscript “Upper Airway Stimulation for Obstructive Sleep Apnea: Durability of the Treatment Effect at 18 months,” which discusses Inspire Upper Airway Stimulation (UAS) therapy, has been accepted for publication in SLEEP.
The UAS therapy durability data demonstrates that significant reductions in obstructive sleep apnea (OSA) severity and quality of life measures were maintained at the 18-month mark. The outcomes data from the 22 center Stimulation Therapy for Apnea Reduction (STAR) Trial cohort demonstrates:
- A 67.4% reduction of apnea-hypopnea index (AHI) at 18 months.
- Quality of life measures including daytime sleepiness and functioning showed clinically meaningful and significant improvement over baseline.
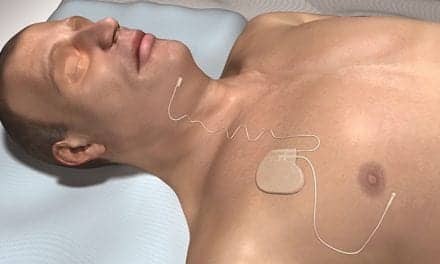
Inspire therapy is an FDA-approved treatment for people with moderate to severe OSA who are unable to use or don’t get relief from CPAP. In contrast to CPAP, Inspire therapy works inside the body and with a patient’s natural breathing process. The system includes a small neurostimulator, a sensing lead, and a stimulation lead. The device is turned on with a handheld Inspire sleep remote and, when activated, Inspire therapy senses breathing patterns and delivers mild stimulation to key airway muscles which keeps the airway open during sleep.
“The clinical evidence supporting the use of Inspire therapy is very strong. With the publication of the 18-month durability data, there are now 9 peer-reviewed publications covering the safety, effectiveness and durability of Inspire therapy,” says Tim Herbert, CEO of Inspire Medical Systems, in a release. “We also expect publication of the 2-year and 3-year outcomes data in the second half of 2015.”
In addition, 2-year and 3-year objective and subjective outcomes data from the STAR Trial will be featured during several scientific sessions at the American Academy of Sleep Medicine (AASM)’s SLEEP 2015 meeting in Seattle, June 6-10, 2015. SLEEP attendees can learn more about Inspire therapy and the recent research results during the following conference presentations in Seattle:
- Tuesday, June 9th:
o S09: “Randomized Trials of Surgery for OSA”
§ Room 6C; 10:20 AM – 12:20 PM
o O19: “Obstructive Sleep Apnea Treatment”
§ Room 6B; 4:30 PM – 4:45 PM - Wednesday, June 10th:
o D04: “How Do New Clinical and Consumer-Oriented Tools Fit Within the Practice of Sleep Medicine?”
§ Room 6B: 8:00 AM – 10:00 AM