The Australian Therapeutic Goods Administration has approved Inspire Medical Systems Inc’s upper airway stimulation device to treat moderate to severe obstructive sleep apnea (OSA) in patients who are unable to benefit from CPAP.
“This approval for our Inspire therapy is the first step to providing patients and physicians in Australia with a new alternative for the treatment of moderate to severe OSA,” says Tim Herbert, president and CEO of Inspire Medical Systems, in a release. “Inspire therapy is supported by a strong and growing body of clinical evidence demonstrating the safety and efficacy of our therapy. We look forward to completing the reimbursement process in Australia and initiating our commercial activity.”
Concurrent with the regulatory review, Inspire has applied for reimbursement in Australia, and the application is under review by the Medical Services Advisory Committee, which is an independent committee established by the Australian Government Minister for Health to evaluate new medical services proposed for public funding. Inspire will continue with the active discussions with the Medical Services Advisory Committee, including in-person meetings in late 2020. The reimbursement process is anticipated to be completed during 2021 and Inspire expects to launch its therapy in Australia thereafter.


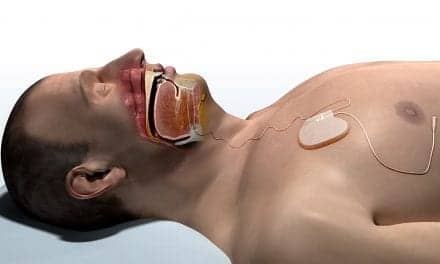

How on earth this is sprouted as minimally invasive is beyond me. Just ask surgeons who do not get paid by Inspire to install the devices how invasive they would see this procedure. There have been issues in the US and people have been silenced. Australians beware.