September 6, 2006
Health Canada has approved sale of St Paul, Minn-based, Restore Medical’s Pillar® Palatal Implant System, providing Canadians access to the FDA-cleared palatal implant to treat snoring and mild-to-moderate obstructive sleep apnea (OSA). The Pillar System is being distributed in Canada through a partnership with Southmedic of Ontario. For the millions of Canadians who are frustrated with traditional treatment options or risky, painful surgery, the Pillar Procedure offers a safe and effective in-office treatment with minimal pain and fast recovery, according to a company press release.
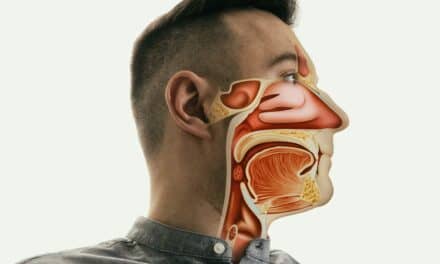
During the Pillar Procedure, a physician places three small polyester implants in the soft palate located at the back of the roof of the patient’s mouth. Airflow in the back of the mouth can cause loose palatal tissue to flutter or vibrate causing snoring, or can collapse and obstruct the upper airway causing OSA. The Pillar implants are designed to add structural support and stiffen the soft palate tissue, thereby reducing or eliminating this tissue flutter, vibration or collapse.